They were the first lasers to operate in the green and blue regions of the spectrum, and some versions provide ultraviolet output. Silfvast, in Encyclopedia of Physical Science and Technology (Third Edition), 2003 VI.A.2 Argon and Krypton Ion LasersĪrgon and krypton ion lasers were discovered shortly after helium–neon lasers. Krypton difluoride is a powerful oxidizing agent and is capable of oxidizing and fluorinating xenon gas to XeF 6 and gold metal to AuF 5. No other molecular fluoride of krypton is known, so all krypton compounds are derived from KrF 2. Although the first krypton compound to be prepared was described as the tetrafluoride, the properties ascribed to this material have been shown to be those of the difluoride. The heat of atomization for KrF 2 is only 97.8 kJ mol −1 and is substantially less than that of F 2 (157.7 ± 0.4 kJ mol −1), making it a better low-temperature source of fluorine atoms and an aggressive fluorinating agent at even low temperatures. Consistent with its thermodynamic instability, krypton difluoride is a powerful oxidative fluorinating agent and is capable of oxidizing xenon to XeF 6 and gold to AuF 5. The standard enthalpy of formation (derived from calorimetric measurements of the gaseous compound at 93 ☌) is 60.2 kJ mol −1. The KrF 2 molecule has been shown, like XeF 2, to be linear in the gas phase, in the solid state, and in solution. It can, however, be stored for indefinite periods of time at −78 ☌. The simplest of these compounds is KrF 2, a colorless crystalline solid which can be sublimed under vacuum at 0 ☌ but is thermodynamically unstable and slowly decomposes to the elements at ambient temperatures. 
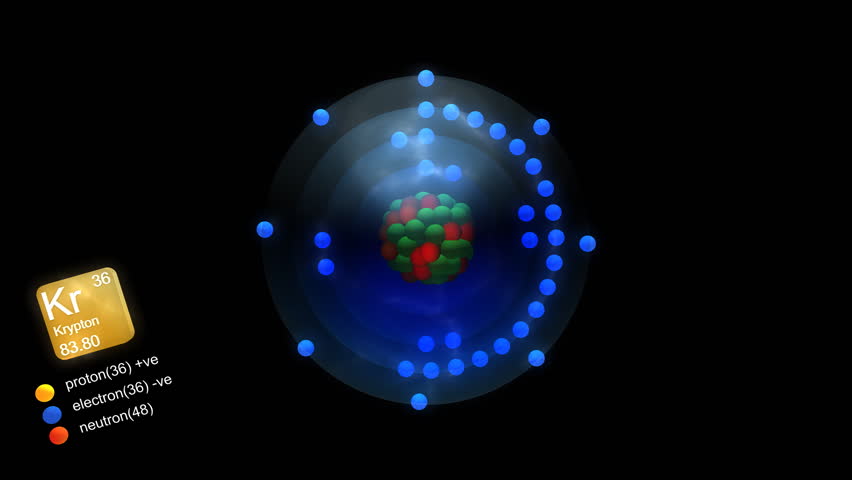
Krypton is the lightest of the noble gases that forms isolable chemical compounds in bulk amounts. Schrobilgen, in Encyclopedia of Physical Science and Technology (Third Edition), 2003 IV.A Krypton Difluoride This is because catalysis is a phenomenon which occurs on surfaces and such clusters will come in a large variety of structures, all with high surface areas.Gary J. If metal clusters show the same kind of behaviour, they may give rise to new types of catalytic activity. The chemists also want to find evidence for similar changes in structure in clusters of metal atoms. ‘One of our long-term objectives is to search for such a transition.’ ‘At some stage in their development towards the bulk,’ says Stace, ‘the clusters will have to change symmetry.’ He and Lethbridge think that this change probably happens in clusters which contain about 2000 atoms. When the inert gases become solids, they always have a structure which crystallographers refer to as face-centred cubic. One of the interesting things about such structures, says Stace, is that there is no equivalent structure for the bulk material. The clusters Kr 147 + and Kr 309 + contain three and four closed shells.Ĭlusters have icosohedral symmetry – that is, 20 regular faces. The magic numbers simply correspond to the numbers of atoms needed to complete closed shells of increasing size. Structures with a closed shell are thought to be particularly stable. Further atoms are then added until the core is completely surrounded by an outer layer, which forms a closed ‘shell’. According to the theory, a cluster grows around a small ‘core’, consisting of a few atoms. The theory to explain why some cluster sizes are favoured is based on a simple picture of the way in which the atoms in a cluster pack together. In their mass spectra, Lethbridge and Stace did indeed see intense peaks which corresponded to Kr 147 + and Kr 309 +. These numbers are also known as ‘magic number combinations’. The theory, which is based on three-dimensional structures known as Mackay icosahedra, predicts that favoured clusters should contain 13, 55, 147, 309, 561, and 923 atoms. Lethbridge and Stace needed a computer to unravel the complications.Ĭhemists have a theory which predicts which sizes of cluster should be favoured. For example, for the cluster ion Kr100+, there are more than 100 million different permutations of the isotopes. The various possible combinations of the isotopes give rise to many extra peaks in the mass spectra.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
